  
For example, under three-fold symmetry, both 4- and 5-coordinate species can accommodate M(E) species with diverse d-electron configurations ( Figure 2). This provides a distinct electronic structure that may be able to accommodate a higher number of d-electrons, so long as those electrons do not fill strongly destabilized orbitals. A third mode of achieving electronic stabilization for multiply bonded species is to remove donor ligands from the central metal under consideration. Such a scenario has been used, for example, to offer an explanation for the unanticipated stability of the d 6 L 5Pt(O) species prepared by Hill and coworkers. Synergism in this context can in principle, via orbital mixing, serve to stabilize d-electrons that would otherwise be destabilized via a strong π* interaction. A second means of achieving electronic stability in M(E) species constitutes using a combination of ligands that are both σ donating and π-accepting. 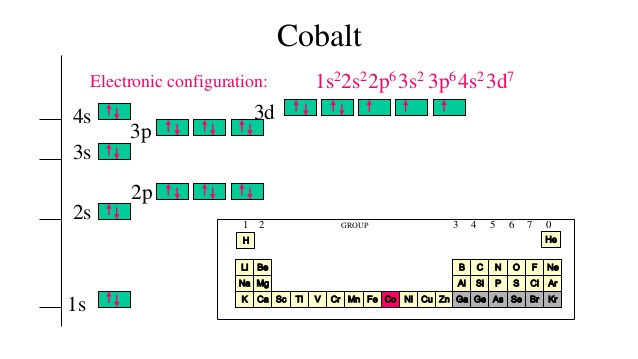 
The high stability of complexes such as trigonal bipyramidal (TBP) V(O) and tetrahedral ( iPr 2N) 3Cr(N) drives home this point. Such an approach is of course not limited to complexes of four-fold symmetry. To a first order approximation, the π-bond order decreases from 2 to 3/2, 1, and ½ as the d 3, d 4, and d 5 configurations are respectively populated. It is therefore not surprising that high-valent early transition metals that feature d 0, d 1, or d 2 electron counts have historically dominated the literature of terminal L 5M(E) complexes. For a prototypical 6-coordinate metal center that features one terminal multiply bonded ligand L 5M(E), the σ* and π* bonding interaction results in the destabilization of four orbitals of d-parentage. The most straightforward and hence historically most familiar way is to preserve a low d-electron count at the metal. With this generalization in mind, there are at present three well-identified pathways to achieve a reasonable degree of electronic stability in an M≡E or M=E interaction (also abbreviated as M(E)). As a consequence, complexes of the type L nM(E) should feature bona fide multiple bond character, and hence electronic stability, when M(E) π* orbitals are vacant or only partially filled, regardless of local symmetry.Įxamples of M≡E and M=E species, with year of publication noted. This MO picture stressed a large splitting of the t 2 g orbitals (Δ π≈ 13,000 cm -1), arising from the presence of a strongly π donating oxo ligand, and the presence of a V≡O triple bond. The molecular orbital (MO) picture of complexes of the type C 4 v-L 5M(E) was developed in the classic study of the d 1 vanadyl ion (VO 2+) by Ballhausen and Gray ( Figure 1). The diverse multi-electron reactivity exhibited by such species is in part attributed to their multiple bond character, and there has hence been much interest in understanding the electronic structures and reactivity patterns of such species. These transformations include, for example, olefin epoxidation and aziridination, C-H bond oxygenation and amination, and e -/H + transfer processes related to nitrogen fixation. Multiply bonded species of the late transition metals (M≡E and M=E, where E = O, N, NR, CR) have been postulated as key intermediates in many synthetic and enzymatic transformations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
